 For example, the orbital angular quantum number \(l\) can never be greater or equal to the principal quantum number \(n(l < n)\). Report Error Is there an error in this question or solution Q II. Because most atoms (hydrogen being the only exception) were.  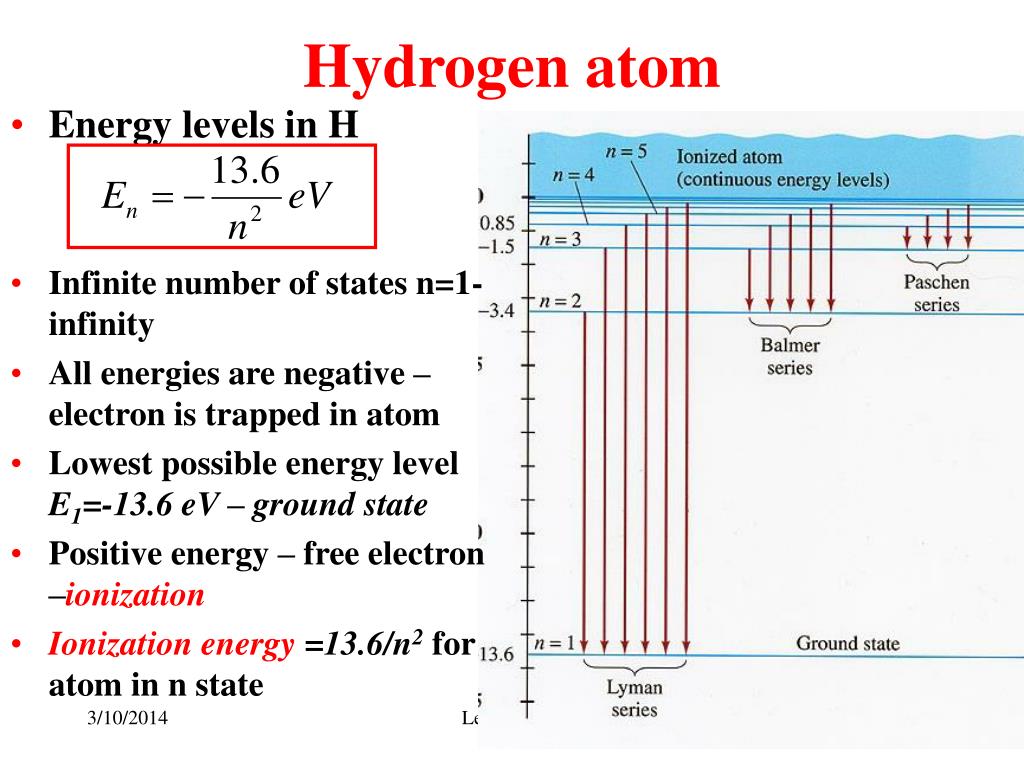
Not all sets of quantum numbers (\(n\), \(l\), \(m\)) are possible. The average atomic mass of hydrogen is 1.008 amu. The proton was found to weigh 1.6726 × 10-24 grams or about 1,836 times as much as an electron. For example, to determine the atomic mass of oxygen, he used the fact that 1 gram of hydrogen reacts with 8 grams of oxygen to make water. 3 Li Lithium 6.94 4 Be Beryllium 9.0122 5 B Boron 10.81 6 C Carbon 12.011. You will need to refer to a periodic table for proton values.(\theta)\Phi_m(\phi). In 1803, English scientist John Dalton published an article in which he assigned hydrogen a weight of 1, and then used compounds of hydrogen to determine the relative weights of the other elements. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. Hydrogen has three isotopes that do not weight he same. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Because hydrogen has one proton and one electron and the 1.0079 is the weighted mass of them both. The "A" value is written as a superscript while the "Z" value is written as a subscript. For an element, relative atomic mass is the average mass of the naturally occurring. (Most of the mass of the universe, however, is not in the form of chemical-element type matter, but rather is postulated to occur as yet-undetected forms of mass such as dark. The electrons have very less mass in comparison to protons or neutrons so the mass of electrons is not influenced in the calculation. Hydrogen, as atomic H, is the most abundant chemical element in the universe, making up 75 percent of normal matter by mass and more than 90 percent by number of atoms. Hydrogen, as atomic H, is the most abundant chemical element in the universe, making up 75 percent of normal matter by mass and more than 90 percent by number of atoms. Isotope, Mass, Abundance, Spin, Mag Moment. 
With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. 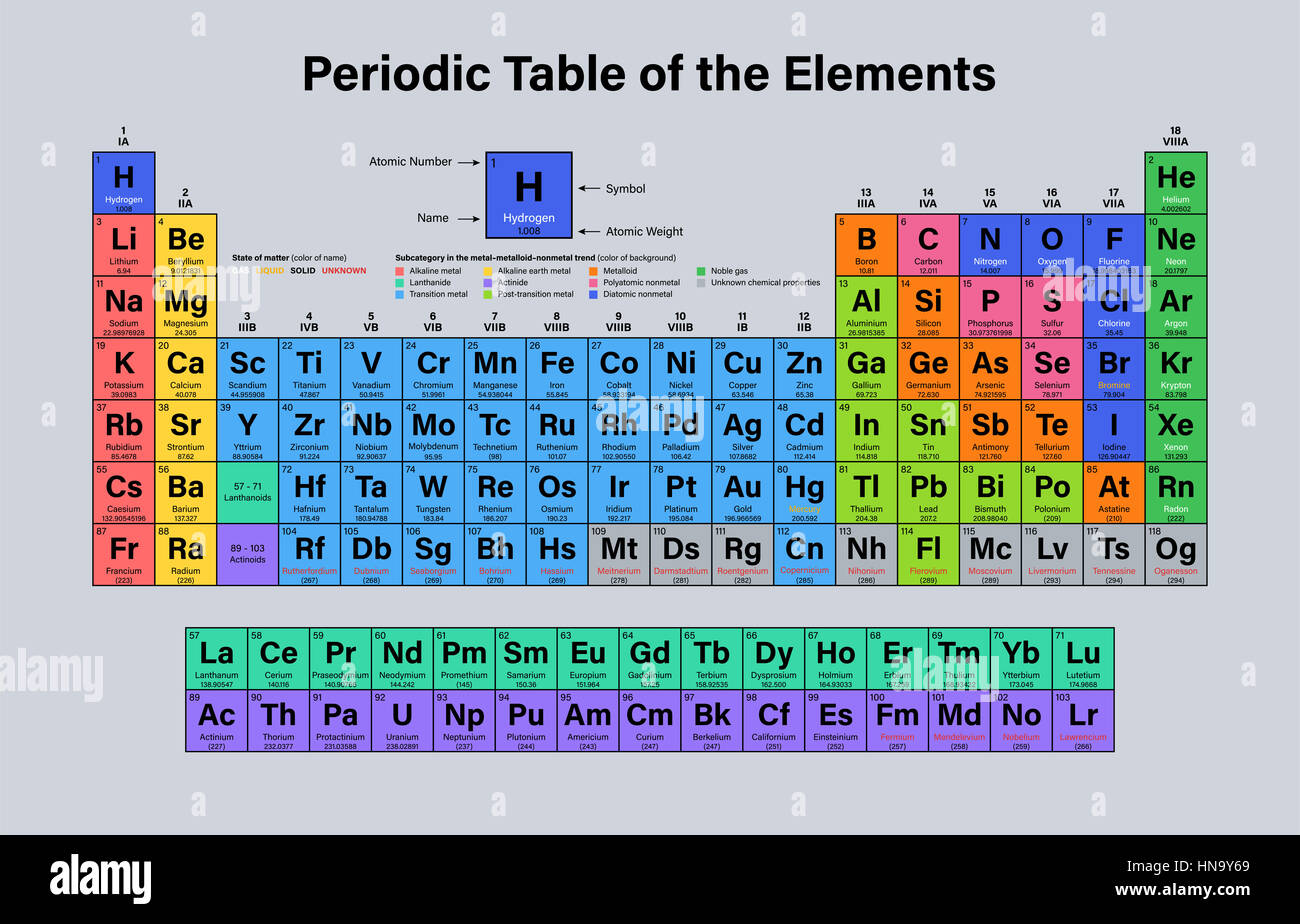
Both the atomic number and mass are written to the left of the chemical symbol. Atomic mass in an atom or group of an atom is the sum of the masses of protons, neutrons and electrons. Hydrogen Atomic Data Hydrogen Strong Lines Hydrogen Neutral Atom. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
